Craig McCotter, MD, FACC, FHRS, with Wilmington Health Cardiology in Wilmington is serving as the lead investigator for the first activated U.S. clinical study site in a new national trial evaluating a potential treatment for Long QT Syndrome Type 2. The study, sponsored by Thryv Therapeutics, will evaluate the investigational medication THRV-1268 in adults living with this rare inherited heart rhythm disorder. Participation in the study reflects Wilmington Health’s ongoing commitment to advancing cardiovascular research and expanding access to innovative therapies for patients in the region. Details about the trial, known as the Wave II Phase 2/3 clinical study, are outlined below.
Thryv Therapeutics Launches Pivotal Wave II Phase 2/3 Study in Long QT Syndrome Type 2
- Wave II clinical study will evaluate THRV-1268, Thryv’s oral SGK1 inhibitor, in adults with Long QT Syndrome Type 2.
- Wave II is informed by the successful Wave I clinical study with LQT-1213 and reinforced by significant QTcF reductions observed in a subsequent clinical study with THRV-1268.
- Individuals and families affected by Long QT Syndrome are encouraged to follow study progress, share awareness, and learn about participation opportunities via Thryv’s social media channels and at https://www.wavestudies.com/wave-2.
Montreal, Quebec – January 13, 2026 – Thryv Therapeutics Inc., a clinical-stage biotechnology company advancing novel serum glucocorticoid inducible kinase 1 (SGK1) inhibitors for inherited cardiac arrhythmias, cardiometabolic diseases, and cardiomyopathies, today announced the initiation of the Wave II clinical study, a pivotal Phase 2/3 clinical research study in adults diagnosed with Long QT Syndrome Type 2 (LQTS2). Participation in Wave II offers an important opportunity to advance scientific understanding and accelerate the development of new treatment options for people and families affected by Long QT Syndrome.
“Wave II represents an important step in advancing THRV-1268 toward a potential approved therapy for Long QT Syndrome,” said Dr. Amy Sehnert, Chief Medical Officer, Thryv Therapeutics. “This rare population lives with a serious and potentially life threatening condition and has limited treatment options, with no FDA-approved therapies specifically indicated to directly treat Long QT Syndrome by addressing QT prolongation. We look forward to advancing THRV-1268 in this late-stage trial and further evaluating its potential as a therapeutic option for patients.”
Thryv’s Wave II study builds on the successful completion of the Wave I clinical program (NCT05906732), as well as supportive findings from a more recent Phase 1 clinical study with THRV-1268 (NCT07186946), which together demonstrated the QT-shortening potential of SGK1 inhibition in acquired and congenital Long QT Syndrome Types 2 and 3. In Wave I, treatment with Thryv’s SGK1 inhibitor, LQT-1213, produced clinically meaningful reductions in QTc across patients with LQTS Types 2 and 3, including on top of standard-of-care therapies, while maintaining a favorable safety profile. When evaluating either the regulatory aligned endpoint of change in area under the curve (AUC) corrected QT interval or the typical clinical measure of maximal change before and after treatment, the magnitude of change on day 3 of treatment was statistically significant and clinically meaningful. These results provided clinical proof-of-concept that inhibiting SGK1 can address the underlying electrophysiologic drivers of QT prolongation.
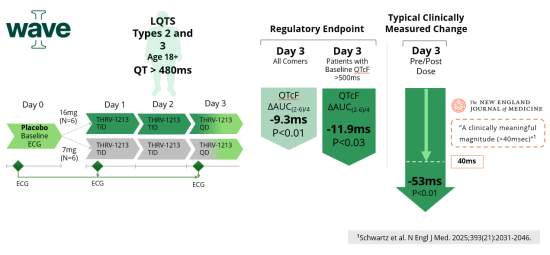
Wave II is designed to evaluate the safety and efficacy of THRV-1268, an orally administered SGK1 inhibitor, and reflects Thryv’s continued progress toward developing a first-in-class disease-modifying therapy for people living with LQTS2 – a rare inherited condition with no currently approved treatments – that directly addresses the underlying QT prolongation. Guided by results from the Wave I study, Wave II is a two-part study comprising a 12-week dose escalation phase (Part A) followed by a 6-week fixed-dose phase (Part B) with a selected dose of THRV-1268. The agreed primary efficacy endpoint is the mean change from baseline in QTc AUC over a given time interval in patients with genetically confirmed LQTS Type 2 with baseline QTc >480 msec.
In parallel, Thryv’s myQTwave non-interventional study (NCT07075445), continues to enroll individuals with LQTS2 and LQTS3 to further advance understanding of the disease and patient experience. Learn more at www.myQTwave.com.
U.S. Site Network and Enrollment Timing
Wave II will recruit across a network of clinical study sites throughout the United States with the first site activated led by Dr Craig McCotter, FACC, FHRS, at Wilmington Health in
Wilmington, North Carolina. Additional U.S. sites are expected to be activated over time as recruitment expands. Study details are available on ClinicalTrials.gov (NCT07277582) and at www.wavestudies.com/wave-2.
About THRV-1268
Thryv’s extensive non-clinical and clinical research has established strong evidence supporting SGK1 inhibition as a therapeutic approach for Long QT Syndrome (LQTS). This foundational work, generated through the company’s first compound, LQT-1213, has enabled the advancement of THRV-1268 as a best-in-class, optimized SGK1 inhibitor designed for long term therapy in this chronic, potentially life-threatening disease. THRV-1268 is an investigational, orally administered SGK1 inhibitor being evaluated in the Wave clinical trials for its potential to reduce QT prolongation and support more stable heart rhythms in individuals with LQTS, including LQTS2.
About Long QT Syndrome Type 2
Long QT Syndrome Type 2 (LQTS2) is a rare inherited cardiac arrhythmia disorder, characterized by delayed ventricular repolarization, which is reflected as prolongation of the QT interval on electrocardiogram (ECG). QT prolongation increases the risk of potentially life threatening ventricular arrhythmias, including torsades de pointes and sudden cardiac arrest. Individuals living with Long QT Syndrome face an ongoing risk of cardiac events, and there are currently no approved disease modifying therapies.
About Thryv Therapeutics
Thryv Therapeutics Inc. is a privately owned company based in Montreal, Quebec, Canada. Thryv is pioneering a precision medicine approach to treat genetic arrhythmia syndromes and cardiometabolic diseases with potent and highly selective inhibitors of serum glucocorticoid inducible kinase 1 (SGK1). For more information, please visit www.thryvtrx.com.
Media Inquiries:
brittany@thryvtrx.com | 514 973 0915
